Category: Asthma
-
Could We “Turn Off” Allergies With Gene Editing? Here’s What CRISPR Actually Makes Possible
A baby with a fatal metabolic disorder was successfully treated with CRISPR gene editing, raising hopes for managing allergies. However, allergies involve complex genetic and environmental factors, making CRISPR a challenging solution. The future may include targeted treatments for immune disorders, but widespread applicability for allergies remains uncertain and ethically debated.
-

How Weight-Loss Drugs and AI Are Reshaping Allergy Medicine
In 2026, GLP-1 drugs and AI are transforming allergy care. While GLP-1 medications alter appetite signaling and aid weight loss, the potential for allergic reactions complicates patient experiences. AI enhances diagnostics by streamlining processes and improving accuracy, ultimately leading to better outcomes and fewer misdiagnoses in allergy treatments.
-

Women, Hormones, and Allergies: Why Your Symptoms Change and What to Do About It
Allergies exhibit different patterns in women and men, largely influenced by hormones. After puberty, estrogen increases allergy risk, causing symptoms tied to menstrual cycles and pregnancy. Understanding hormonal patterns can help women manage these flares with proactive treatment strategies, rather than reactive responses, improving overall quality of life through various life stages.
-
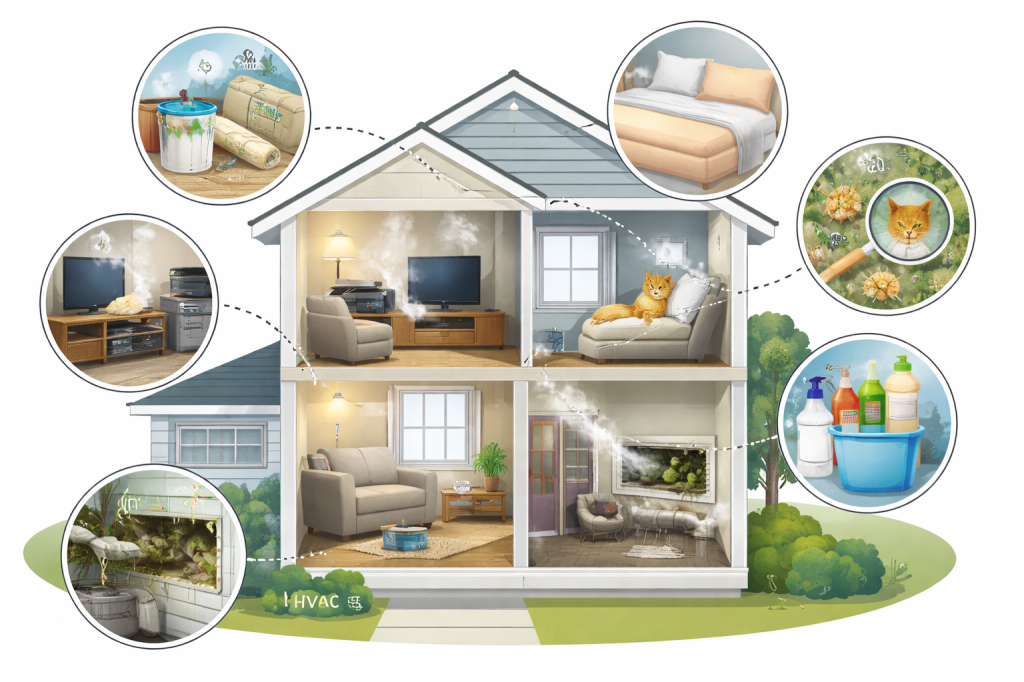
Hidden Allergens in the Home: What’s Really Irritating Your Body
Allergies are often underestimated as indoor problems, with volatile organic compounds (VOCs) and mold being significant irritants. VOCs are released from everyday items like carpets and electronics, while mold proliferates in HVAC systems. Both contribute to chronic respiratory issues, prompting the need for effective measures to improve indoor air quality and humidity management.
-

Hidden Allergens in Popular Diets: What You’re Overlooking (and How to Fix It)
The shift toward diets like keto, vegan, and paleo introduces hidden allergens that can pose serious risks. Reformulations in packaged foods, cross-contact during food preparation, and mismanagement of allergies complicate safety. It’s essential to be vigilant about ingredient changes and maintain a food diary to distinguish between true reactions and dietary adjustments.
-

The Allergy Crisis No One in Public Health Wants to Talk About
Allergy rates have risen dramatically in the past 30 years, largely due to the hygiene hypothesis, which limits immune system exposure to microbes. Climate change also worsens allergies, while socioeconomic factors complicate the issue. Public health efforts prioritizing education, local pollen counts, and preventive measures could significantly reduce allergy-related healthcare costs and improve outcomes.
-

The Future of AI in Allergy Care: How It’s Already Changing Everything
AI is revolutionizing allergy care by significantly reducing the time allergists spend on documentation and enhancing diagnosis support. It improves the accuracy of medical records, enriches patient visits, expedites pre-screening, and streamlines business operations. Although AI augments physicians’ capabilities, human oversight remains crucial, paving the way for more personalized patient care.
-

The Biggest Breakthroughs in Allergy Research This Year
Most allergy treatments still play defense. You react, you medicate, and then you wait for it to come back. But this year? We’re finally playing offense. From food allergies to eczema to asthma, the latest breakthroughs are revolutionizing the entire field. Here’s what’s new and what you can do about it right now. 1. Biologics…
-

The Role of Vitamin D in Allergy Development and Prevention
Vitamin D deficiency is common and often overlooked in allergy patients, impacting immune functions and potentially worsening allergies. It regulates immune responses and supports gut health. Natural sunlight exposure is key for boosting levels, along with proper supplementation. Regular blood tests are essential for monitoring levels and ensuring optimal health.
-

When Allergies and Eczema Collide: Understanding the Atopic March
The atopic march refers to the progression of allergic conditions in children, typically beginning with eczema or food allergies and potentially leading to asthma. Early detection and intervention, including testing IgE levels, tracking triggers, and consulting an allergist, are crucial to managing these conditions and preventing further complications.